Contract Research Organization (CRO)
"Medconsult Geo" LLC
Über uns




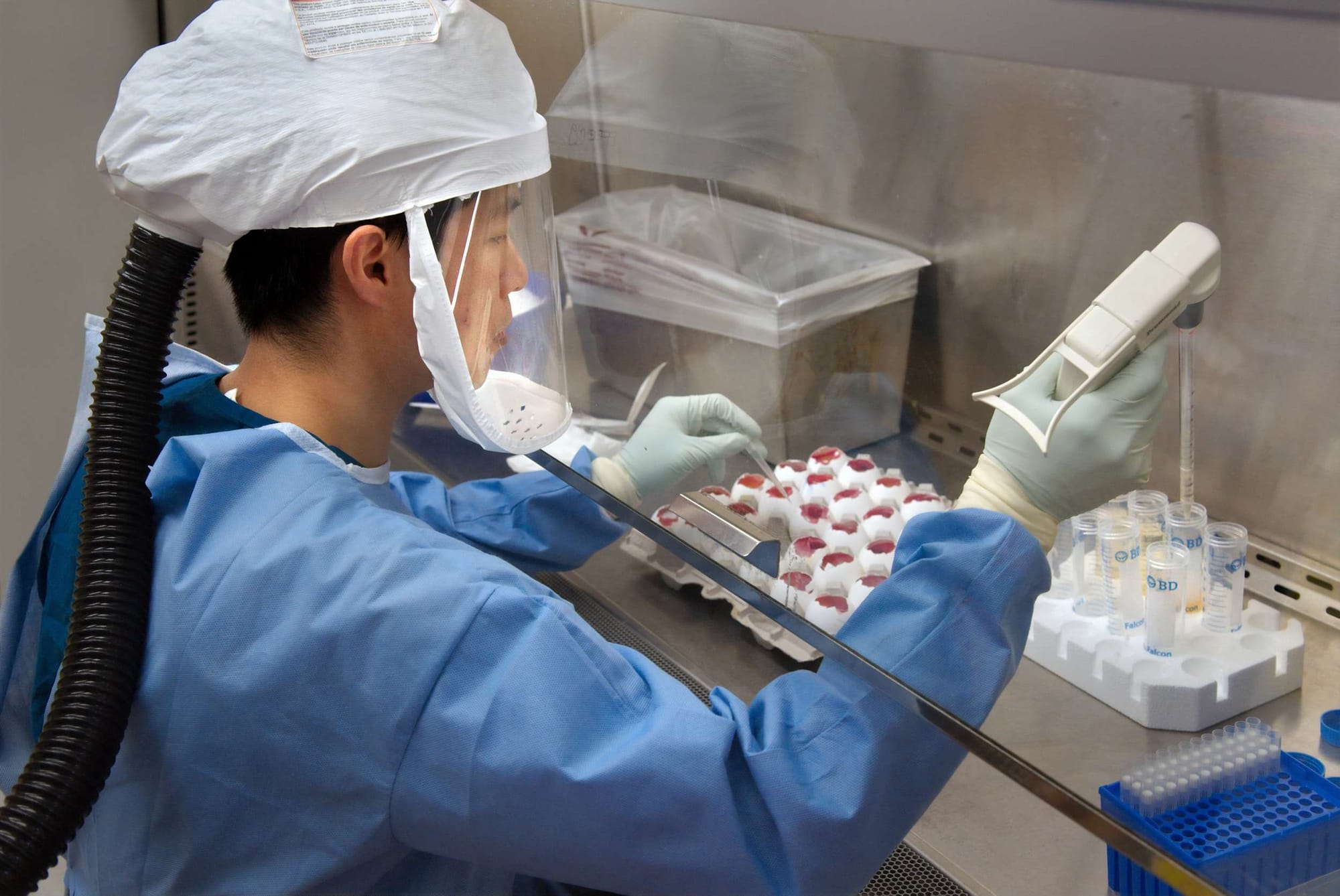
"Medconsult-Geo" LLC is the German-Georgian contract research organization (CRO), which operates successfully in Georgia and cooperates closely with many world-famous biopharmaceutical companies as well as manufacturers of medical devices and products. “Medconsult-Geo” LLC provides support to the pharmaceutical, biotechnology, and medical device industries in the form of research services outsourced on a contract basis. We can provide such services as biopharmaceutical development, biologic assay development, commercialization, preclinical research, clinical research, clinical trials management, and pharmacovigilance.
For about 12 years, with the direct participation of our scientists and colleagues, several international all phases of clinical studies have been carried out and some are currently in progress.
Our goal is to build up close and long-term cooperation with pharmaceutical companies and medical product manufacturers worldwide, to offer them in Georgia, very attractive conditions, both financial and time-saving, for conducting high-quality clinical trials and beyond that, to achieve together scientifical victories in medicine.
What makes Georgia and, in particular, our contract research organization "Medconsult-Geo" LLC such an interesting destination for conducting clinical studies?
1. The preparatory phase for the implementation of clinical studies or the collection of all legally prescribed procedures takes an average of 4 to 5 months.
These procedures are:
- • Obtaining all necessary approvals for clinical trials from the Georgian Ministry of Health and ethics committees.
- • Translation of all necessary documents, first certification and then insertion of these documents into the appropriate legal format;
- • Establishing cooperation with appropriate clinical study centers and researchers.
2. For the "Sponsor" company, the implementation of a clinical study in Georgia is associated with significantly lower financial expenditure (approx. 50% cost reduction) than is to be expected in other highly developed European, Asian or American countries.
3. It is also worth mentioning the very high quality of the clinical studies carried out, which relates to the following:
- • Many years of experience of our employees, as well as colleagues from Georgian research centers;
- • Multiple international certifications of both our CRO-employees and scientists from research centers; (GCP-certificates)
- • The research centers that cooperate with our CRO are mostly equipped with modern equipment;
- • Direct or close cooperation with German and Austrian laboratory research centers, whereby materials for detailed research or further assessment can be sent if necessary;
5. "Medconsult-Geo" LLC is a member of several international marketing companies and medical-biological associations, including:
- The California Life Science Association BIOCOM
- Scientist Marketing Corporation and
- International Society for Stem Cell Research (ISSCR).
We would be delighted if our CRO "Medconsult-Geo" LLC would attract your attention to the implementation of joint clinical studies in Georgia, taking into account our heartfelt wish for long-term cooperation.
I'm looking forward to your response, Sincerely

Director of Company,
Contract Research Organization (CRO)
"Medconsult-Geo" LLC
Dr. jur. Gocha Endeladze

Hauptvorstand
Prof. Dr. med. Dr. hc Reinhold Lang
BeraterProfessor für Allgemeines, Viszeral, Laparoskopie und Roboterchirurgie
Prof. Dr. med. Fuat Oduncu
BeraterProfessor für Onkologie
Prof. Dr. med. Dr. hc Dirk André Cleverts
BeraterProfessor für Radiologie
Prof. Dr.Sci, Ph.D. Tinatin Chikovani
BeraterProfessor für Immunologie
Prof. Dr. med. Markus Michael Suckfüll
BeraterProfessor für Hals-Nasen-Ohren-Heilkunde, Kopf- und Halschirurgie
Prof. Dr. med. Dr. hc George Grigolia
BeraterProfessor für Allgemeine Chirurgie
Prof. Dr. med. Roland Huf
BeraterProfessor für postoperative Versorgung und Notfallmedizin
Prof. Dr. med. Nodar Lomize
BeraterProfessor für Allgemeine und Viszerale Chirurgie
Prof. Dr. med. Bernd Gerhard Lorbeer
BeraterProfessor für Labormedizin
Prof. Dr. med. Horst-Günter Maxeiner
BeraterProfessor für Mikrobiologie, Virologie und Infektionskontrolle
Professor MD. PhD. Zaza Avaliani
BeraterProfessor für Pulmonologie
Prof. Dr. med. Renat Akhmerov
BeraterProfessor für Plastische Chirurgie
Directorate
Dr. Gocha Endeladze
General Director of Contract Research Organization "Medconsult-Geo" LCCProf. Dr. med. Dr. hc George Grigolia
Consultant, Deputy-Director and Coordinator of the main board Contract Research Organization "Medconsult-Geo" LCCProfessor für Allgemeine Chirurgie
Prof. Dr. med. Tamara Tchelidze
Stellvertretender Direktor und HauptberaterScientist and major consultant at Marketing and contact person for the Contract Research Organization "Medconsult-Geo" LCC in USA, Germany, Austria
Mr. Babak Amin
Marketingleiter und Hauptansprechpartner des CRO "Medconsult-Geo" LLC in AsienHauptansprechpartner der Medconsult-Geo LLC in China, Taiwan, Thailand, Südkorea, den Vereinigten Arabischen Emiraten und Saudi-Arabien
Frau Rita Gudadze MD
Berater und klinischer WissenschaftlerBerater für Marketing und Ansprechpartner für die Vertragsforschungsorganisation "Medconsult-Geo" LLC in Kanada und den USA
Frau Mariana Macharadze
Wissenschaftlicher Hauptberater für Management und MarketingWissenschaftlicher Hauptberater für Management und Marketing als Ansprechpartner für die Vertragsforschungsorganisation (CRO) "Medconsult-Geo" LLC in Europa
Dr. Daniel R. Sivette
BeraterBerater bei Marketing und Ansprechpartner für die Auftragsforschungsorganisation "Medconsult-Geo" in der Schweiz und in Frankreich
Frau Ana Macharadze
Wissenschaftlicher Berater für Management und MarketingWissenschaftlicher Berater für Marketing und Management sowie Hauptansprechpartner des CRO "Medconsult-Geo" LLC in Osteuropa, Lateinamerika, Australien und Neuseeland
Herr Arda Fenik
CosultantBerater für Marketing und Ansprechpartner für die Vertragsforschungsorganisation "Medconsult-Geo" in der Türkei, in Aserbaidschan und im Iran
Wissenschaftler
Prof. Dr. Sofio Khardzeishvili MD, PhD
Klinischer Wissenschaftler, General ManagerAssoc. Professor für Gynäkologie und Reproduktologie
Assoc. Prof. Dr. Besarion Lasareishvili
Clinical Scientist and Major Coordinator of the preclinical TrialsPrincipal Investigator, Assoziierter Professor Abteilung für Zelluläre Immunologie
Prof. Dr. Vladimer Papava MD. PhD.
Klinischer WissenschaftlerDoktor der Medizin. Assistenzprofessor, Urologe
Assoc. Prof. Dr. Tamar Svanidze
Klinischer WissenschaftlerAssoc. Professor, Kardiologe an der First Central University Clinic
Assoc. Prof. Dr. med. Tamar Saralidze
Klinischer WissenschaftlerArzt, Kardiologe an der First Central University Clinic
Assistenzprofessor Dr. med. Teimuraz Goletiani. PhD.
Klinischer WissenschaftlerProfessor für Radiologie
MD. Vasil Natsvlishvili
Klinischer Wissenschaftler, BeraterArzt für Herz-Kreislauf-Chirurgie
MD. Lika Injia
Klinischer WissenschaftlerKardiologe am Herz-Kreislauf-Zentrum
MD. Eka Arziani
Klinischer WissenschaftlerArzt, Kardiologe an der First Central University Clinic
MD. Mariam Arabidze
Klinischer WissenschaftlerNephrologe, Doktor der Inneren Medizin und Dialysetherapie
MD. Nino Pochkhua
Klinischer WissenschaftlerMD, Rheumatologe am Science / Practical Center of Rheumatology
MD. Irina Kvikvinia
Klinischer WissenschaftlerMedizin Doktor der Geburtshilfe und Gynäkologie
Frau Tamar Chomakhashvili
Klinischer WissenschaftlerKlinischer Wissenschaftler in der Pharmakologie
MD. Otar Gakhokia
Klinischer WissenschaftlerDoktor der Medizin in Neurochirurg
MD. Zviad Eradze
Klinischer WissenschaftlerArzt für Kardiologie
MD. Sophiko Mebuke
Klinische WissenschaftlerDoktor der Medizin, Endokrinologe,
MD. Nana Egiashvili
Klinischer WissenschaftlerDoktor der Medizin, Kardiologe und Radiologe
Frau Nino Burduli
Klinischer Wissenschaftler, IT-Technische AbteilungMD IRINE UKLEBA
Klinischer WissenschaftlerOrthopäde
MD Lela kupreishvili
Klinische WissenschaftlerDoktor HNO-Arzt
MD. Irma Natsvlishvili
Klinischer WissenschaftlerArzt für Kardiologie
MD. Gocha Tsotsoria
Klinischer WissenschaftlerArzt für Kardiologie
Erfahrung
The Experience of our team with international clinical trials:
1. PI - Phase III A Study the Venous Thromboembolic Risk 2016-2018
2. SC - Phase III Diabetes Mellitus type 2 (2014-2016 current)
3. SI - Phase III COPD treatment study (2014-2015)
4. SI - Phase III pneumonia treatment study, (2014-2015)
5. Phase of the study III, Cardiological Study, Sub investigator (2014-2015)
6. Phase of the study III, Nephrology, Sub investigator (2014-2015)
7. Phase of the study III COPD and Cardiovascular Diseases;
8. Phase of the study III for type 2 Diabetes and Cardiovascular Diseases
9. Phase of the study III, for type 2 Diabetes and Cardiovascular Diseases,
10. Phase III, 1 study in Cardiovascular Diseases
11. Phase III Ponesimod in adult with relapsing multiple sclerosis
12. Phase of the study II, Indication Rheumatology, /2016 – 2018/
13. Phase of the study III, Indication Nephrology, /2017 – 2018/
14. Phase of the study III, Indication Cardiology, /2014 – 2015/
15. Phase of the study III, Indication Nephrology, /2017 – 2018/
16. Phase of the study III, Global Alliance for TB Drug Development 11/2017-12/2021
17. Phase of the study III, International Science and Technology Center (ISTC) 2016-2018,
18. Phase of the study III, randomized controlled multi-center trial, 01/2020-01/2025
19. Phase IV, New Diagnostics, TB- Disease, 2015-2017,
20. Phase IV, Tuberculosis, National Institute of Allergy and Infectious Diseases, 2015-2017
21. Phase of the study III-IV, in tuberculosis. 2013-2018
22. Phase of the study III Human Diagnostic, IC in TB Hospital, 2015 – 2016,
23. Swiss National Fund, Switzerland Georgia, 2009-2012, Disability Status Assessment project
24. Timeline 2014-2015, Phase of the study III, Indication Cardiology, Role Subinvestigator
25. Timeline 2017-2018, Phase of the study III, Indication Nephrology, Role Subinvestigator
26. Timeline 2016-2018, Phase of the study II, Indication Rheumatology, Role Subinvestigator
27. Timeline 2010 – 2013, Phase of the study III, Indication Cardiology, Role Subinvestigator
28. Timeline 2020 -present, Phase of the study II, Indication Rheumatology, Role Subinvestigator
29. Timeline 2019 -present, Phase of the study III, Indication Cardiology, Role Pharmacist
30. Timeline 2017-2018, Phase of the study III, Indication Nephrology, Role Pharmacist
31. Timeline 2014 – 2015, Phase of the study III, Indication Cardiology, Role Pharmacist
32. Timeline 2011-12, Phase of the study III, Indication GOUT, Role Subinvestigator
33. Timeline 2012-2014, Phase of the study II, Indication RA, Role Subinvestigator
34. Timeline 2013-2014, Phase of the study II, Indication RA, Role Subinvestigator
35. Timeline 2014-2015, Phase of the study III, Indication RA, Role Subinvestigator
36. Timeline 2016-2018, Phase of the study II, Indication RA, Role Subinvestigator
37. Timeline 2017-2018, Phase of the study II, Indication RA, Role Subinvestigator
Lokalisierung
Dienstleistungen
Mitgliedschaft
Kontakt
- Ilia Chavchavadze Avenue, 0179 Tbilisi, Georgia
- +49-17621682911
- infocenter@medconsult-geo.com
- Mo-Fr - 08: 00-19: 00 Uhr
Nachrichten
Am Wochenende nutzten Genfer Feuerwehrleute Miniaturtechnologie, um ein asiatisches Hornissennest aufzuspüren.
Mehr lesenWährend einige Drogerien über einen Mangel an Grippeimpfstoffen berichten, ist in den USA eine Rekordzahl an Dosen unterwegs
Mehr lesen